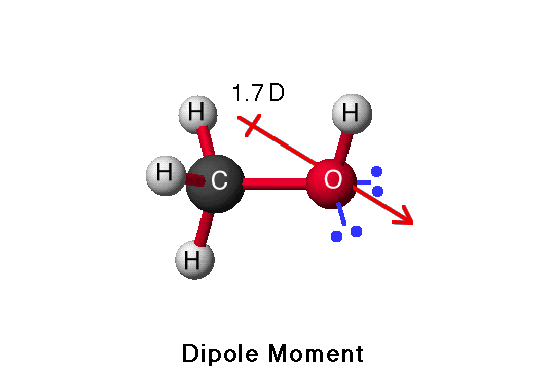
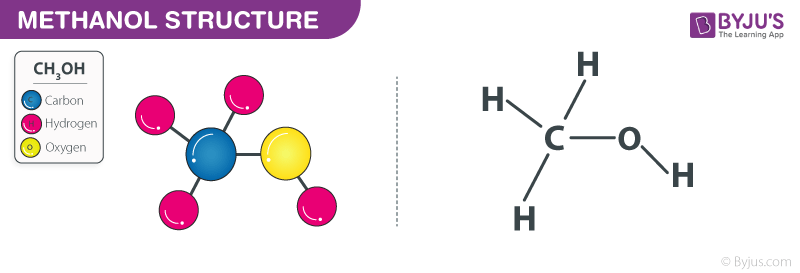
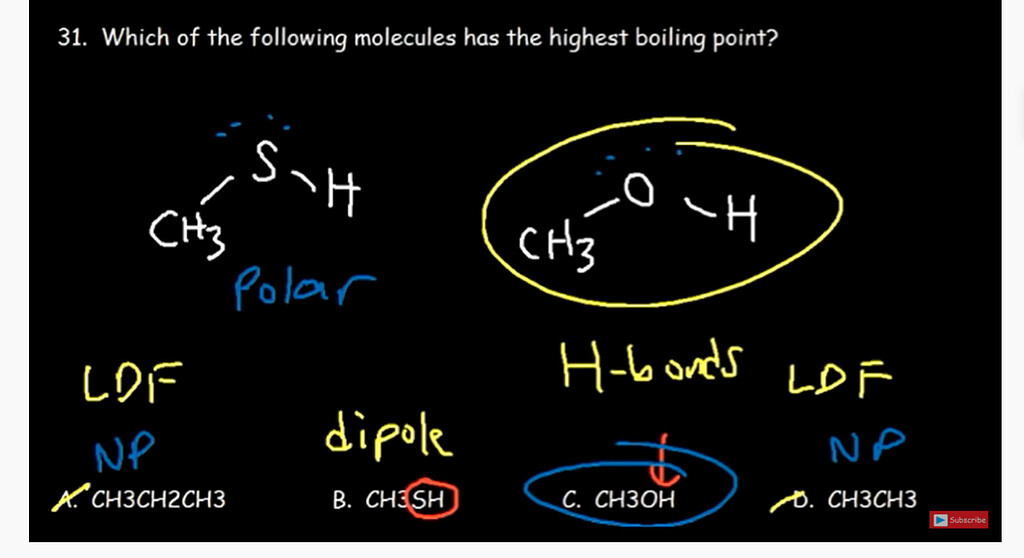
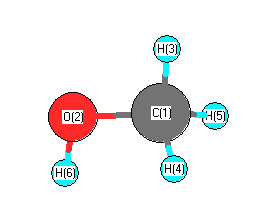
Draw the Lewis structures for methanol (CH3OH) and formaldehyde (H2CO). Use your knowledge of phases and intermolecular forces to predict which liquid would have a higher vapor pressure, boiling point, surface tension,

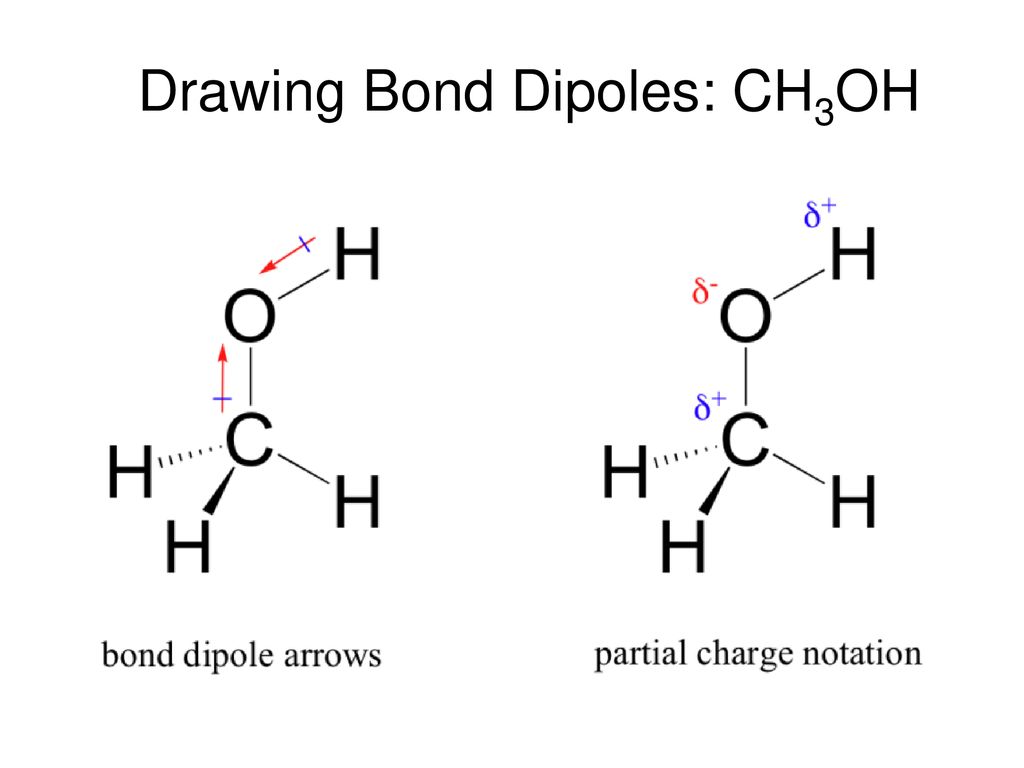
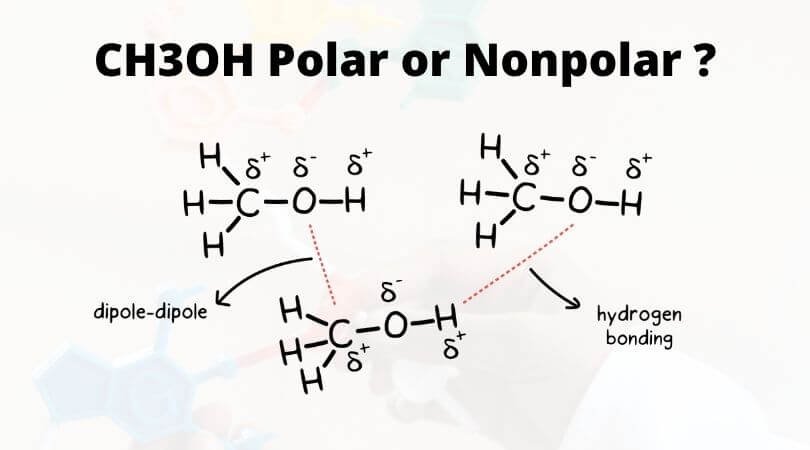
Which drawing best accounts for the polarity of methanol, CH3OH, and the bond polarities that make a major - brainly.com

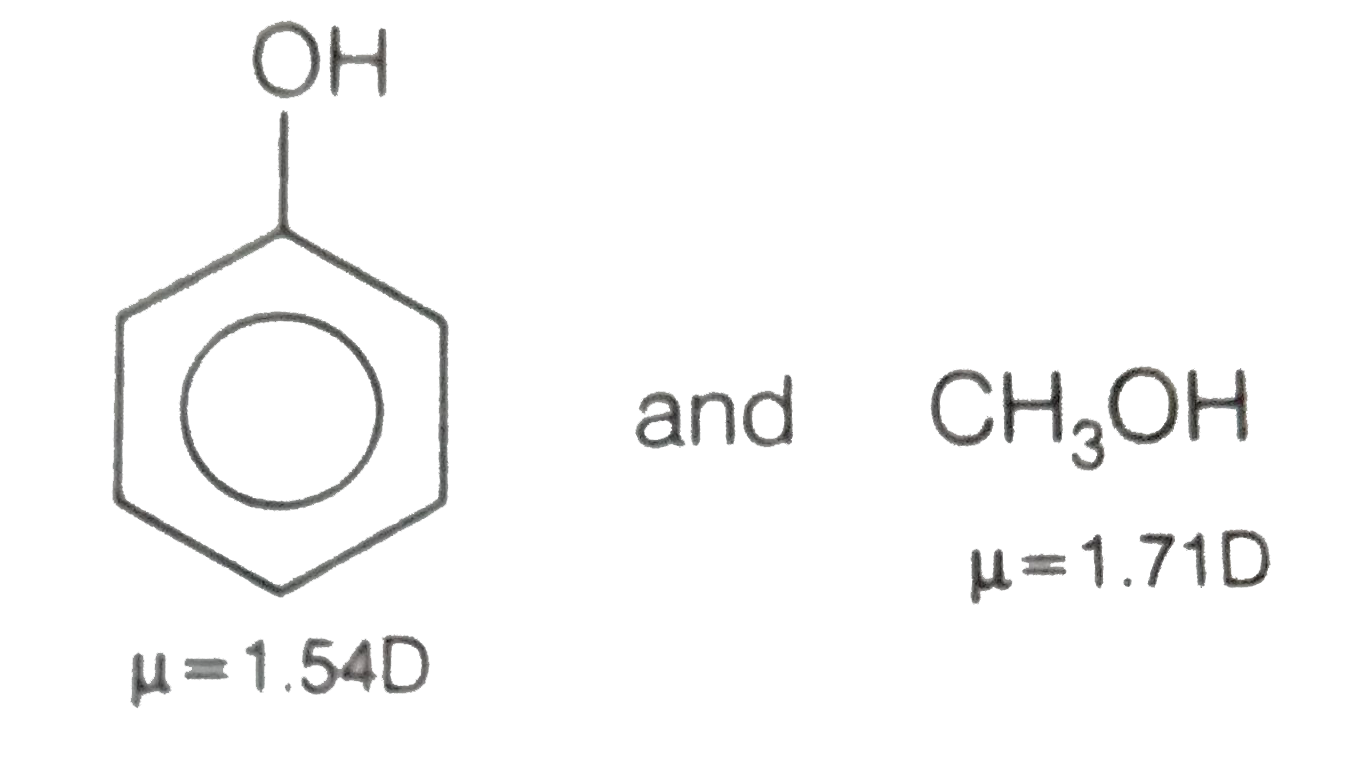
20. Arrange the following in the order given below: a) Decreasing dipole moment : CH3C1, CH2Cl2, CHCI, & CCI. b) Increasing order of dipole moment : CH3OH, CH4, CF., COCHE