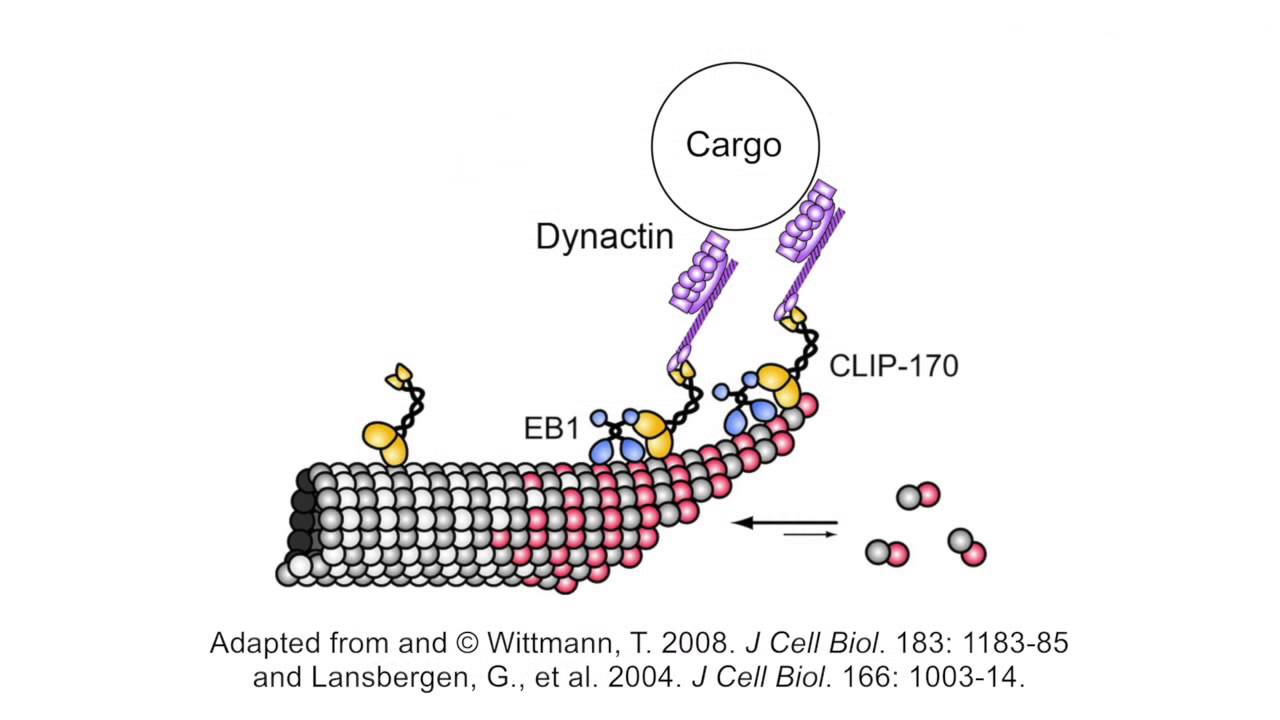
Domain architecture of CLIP-170 and EB1. A, domain architecture of EB1... | Download Scientific Diagram

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology

CLIP-170 is essential for MTOC repositioning during T cell activation by regulating dynein localisation on the cell surface | Scientific Reports
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme
CLIP-170S is a microtubule +TIP variant that confers resistance to taxanes by impairing drug-target engageme
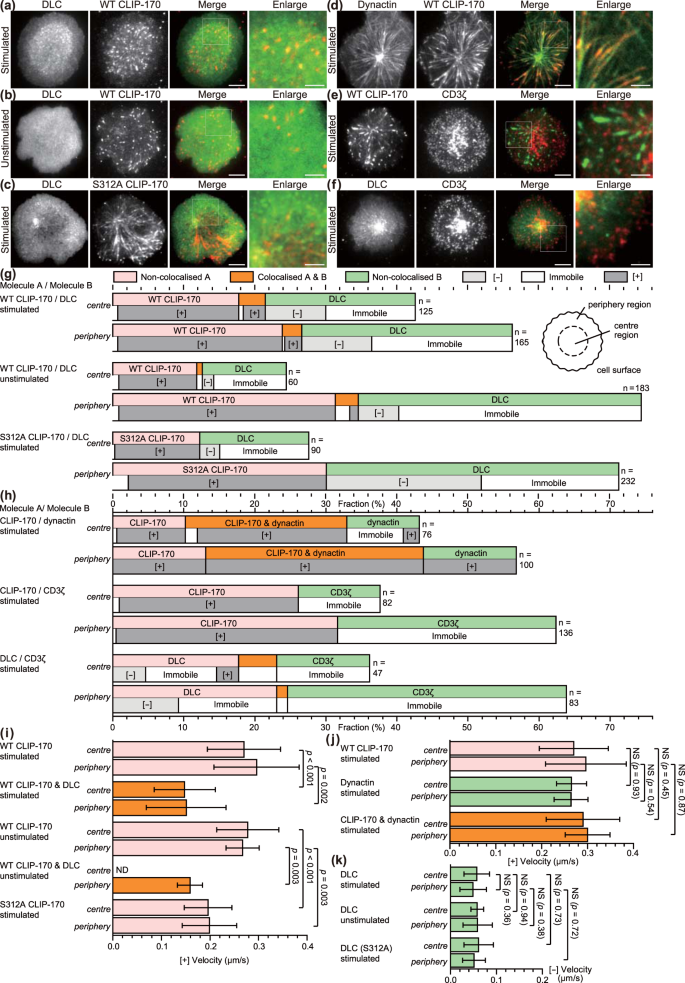
CLIP-170 is essential for MTOC repositioning during T cell activation by regulating dynein localisation on the cell surface | Scientific Reports

α-Tubulin Tyrosination and CLIP-170 Phosphorylation Regulate the Initiation of Dynein-Driven Transport in Neurons - ScienceDirect

Arsenic trioxide disturbs the LIS1/NDEL1/dynein microtubule dynamic complex by disrupting the CLIP170 zinc finger in head and neck cancer - ScienceDirect
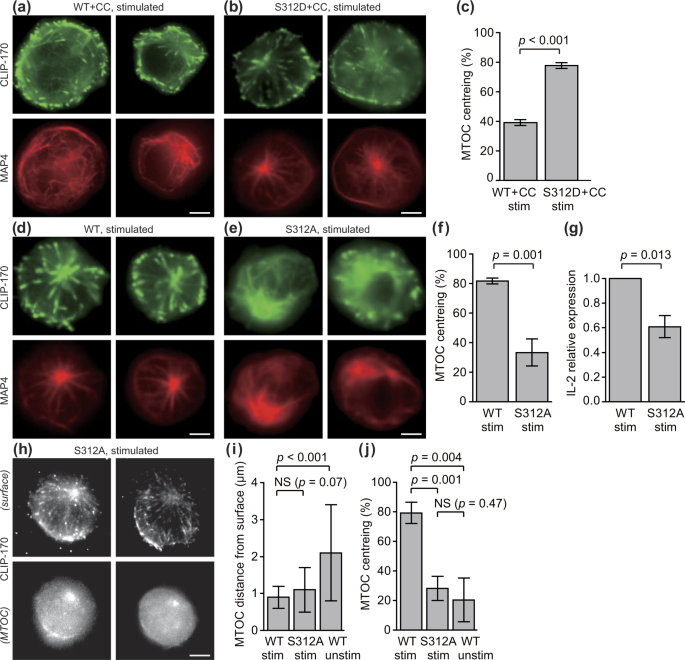
CLIP-170 is essential for MTOC repositioning during T cell activation by regulating dynein localisation on the cell surface | Scientific Reports