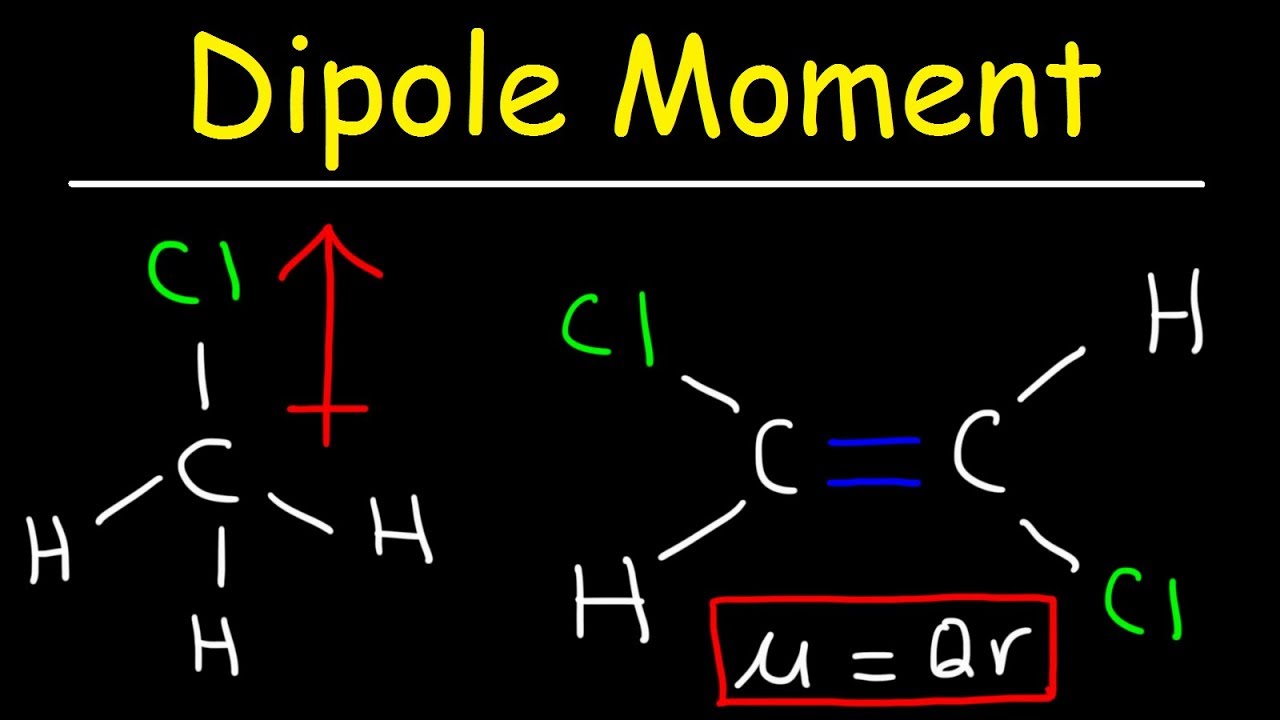
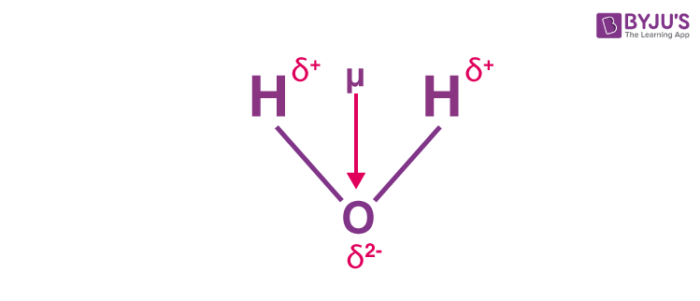
Welcome to Chem Zipper.com......: Dipole moment of CCl4 is zero while that of CHCl3 is non zero. explain.
Which of the following molecules has no dipole moment? (a) CH3Cl (b) CHCl3 (c) CH2Cl2 (d) CCl4 - Sarthaks eConnect | Largest Online Education Community
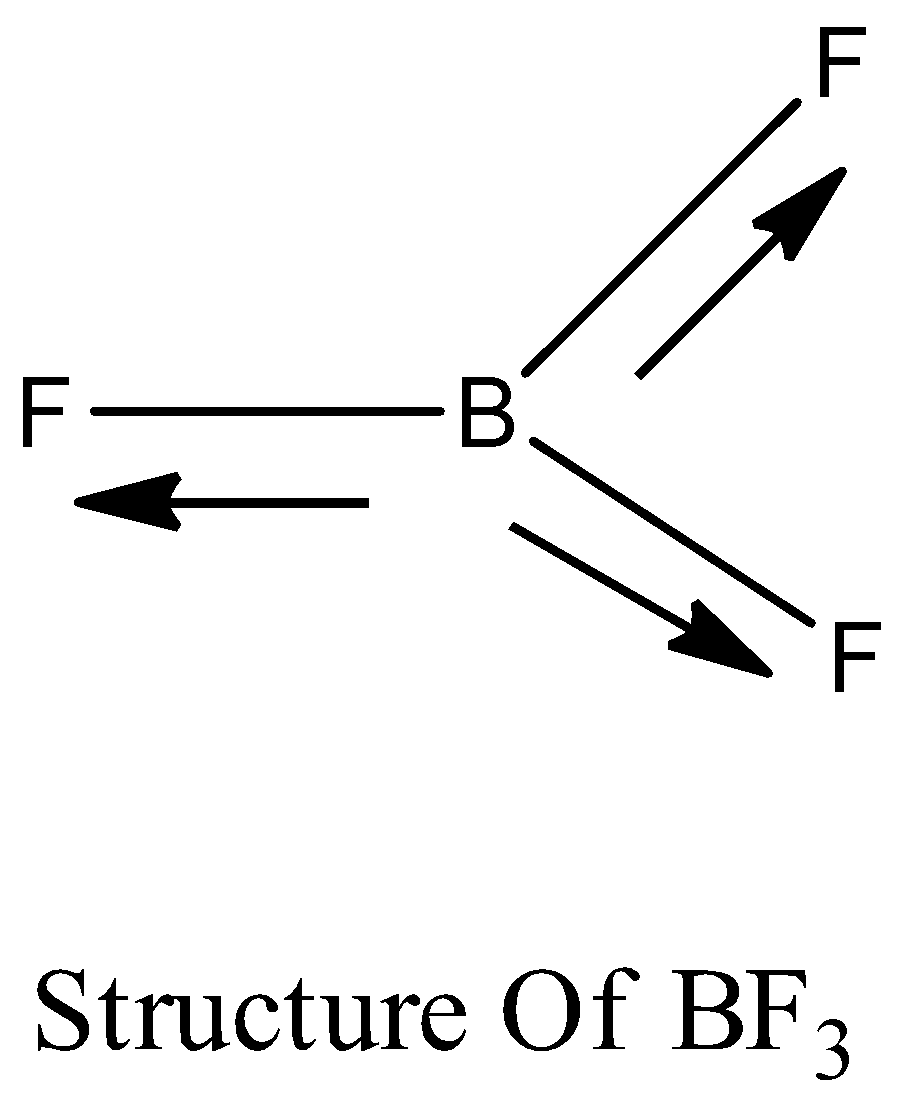
Boron trifluoride $(B{F_3})$ has no dipole moment $(\\mu = 0D)$. Explain how this observation confirms the geometry of $B{F_3}$ predicted by VSEPR theory.


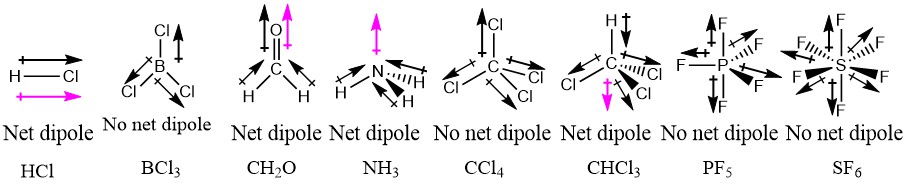
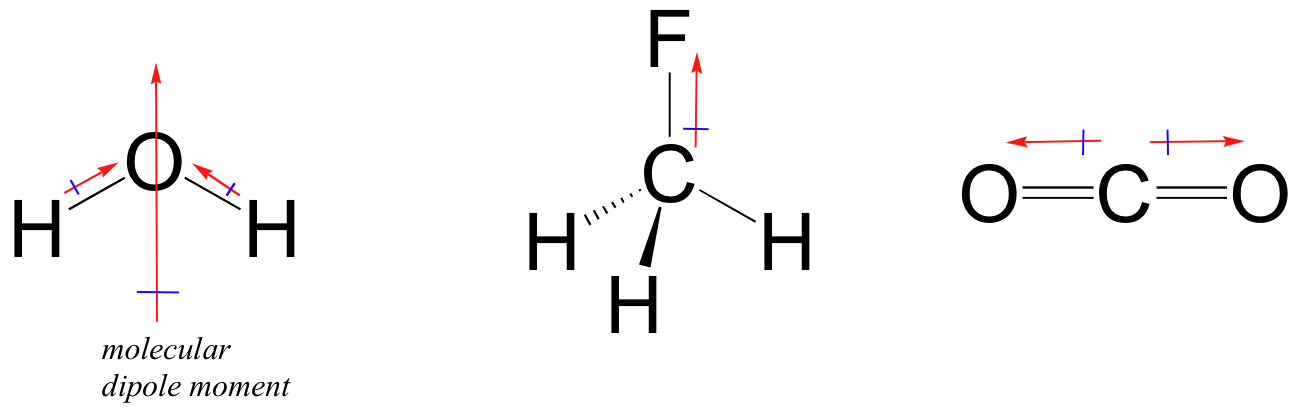

/chapter3/pages3and4/page3and4_files/cnodipole.png)